The human immune system is one of the most complex and fascinating components of the human body. Any time a foreign antigen—whether a pathogen like a virus or bacteria, a parasite or fungi, or an allergen—enters the bloodstream, the immune system springs into action, sending an army of Y-shaped antibodies to work.
Once the body identifies a target, antibodies use compounds to find and bind antigen proteins with their Fab region. The Fc region then binds to a host of other immune compounds that assist in killing the antigen or triggering a chain reaction within the immune system. Antibodies also work to recognize and mark the invading substance, so that the immune system can identify and fight it more quickly in the future. The complex mechanisms of the immune system have been the subject of extensive scientific research since antibodies were first discovered in the 1890s.
New Studies Highlight Critical Information About JAGN1 (Jagunal homolog 1)
Years ago, the University of British Columbia revealed an interesting connection between the JAGN1 gene and the immune system. Now, researchers at the same lab have discovered how the protein associated with JAGN1 participates in producing antibodies. Interestingly, both studies investigated a disease caused by a mutation in the JAGN1 gene.
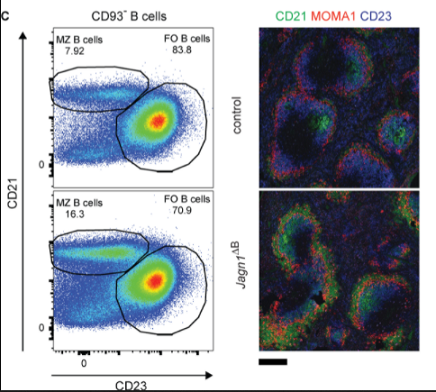
It has been found that patients with this mutation develop a severe congenital neutropenia disease, or SCN, marked by an extremely low level of neutrophils. This white blood cell helps to release enzymes that kill invading antigens and begin ingesting them. Since immune systems cannot destroy these antigens effectively, people with SCN frequently experience severe infections. However, UBC’s study revealed a relationship between JAGN1 and B-cells, the white blood cells capable of transforming into plasma cells when they sense a foreign invader.
B-Cells and JAGN1
When B-cells are functioning as plasma cells, they can produce millions of antibodies within the cell’s endoplasmic reticulum. During the final steps of antibody production, the B-cell ensures that each antibody undergoes glycosylation—or the placement of sugar molecules on the portion of the antibody that interacts with other immune cells. These sugar molecules take on a unique pattern that heavily affects protein stability and the ability of surface proteins to bind to and communicate with other cells and the surrounding environment.
During the UBC study, researchers discovered that the removal of JAGN1 in mice correlated with a remarkable reduction in antibodies produced by the B-cells. Of those antibodies, the glycosylation patterns were drastically different, affecting their ability to bind with proteins and other immune system entities. After replicating the experiment in human tissue with the same results, researchers concluded that the JAGN1 gene has a vital role in the B-cell endoplasmic reticulum’s production of antibodies—as well as an effect on the sugar structures that make protein stability possible.
Looking Ahead
Although SCN is a relatively rare condition, the UBC study demonstrates how careful research of rare diseases can lead to crucial discoveries about how the body works. The same biologic principles are at play each time the human immune system encounters an antigen, and research regarding these rare genetic mutations can help scientists understand the various factors that help them work. In the future, researchers hope to learn more about how to combat disease by unraveling the complexities of the immune system.
Resources:
https://doi.org/10.1084/jem.20200559
*****
From time to time, GraniteGrok accepts content from third parties (posts, or additional links after initial publication) from which we may or may not receive compensation.